Foreign
China’s second Covid vaccine, Sinovac gets WHO emergency approval
Published
3 years agoon
By
Vida Essel-Lamptey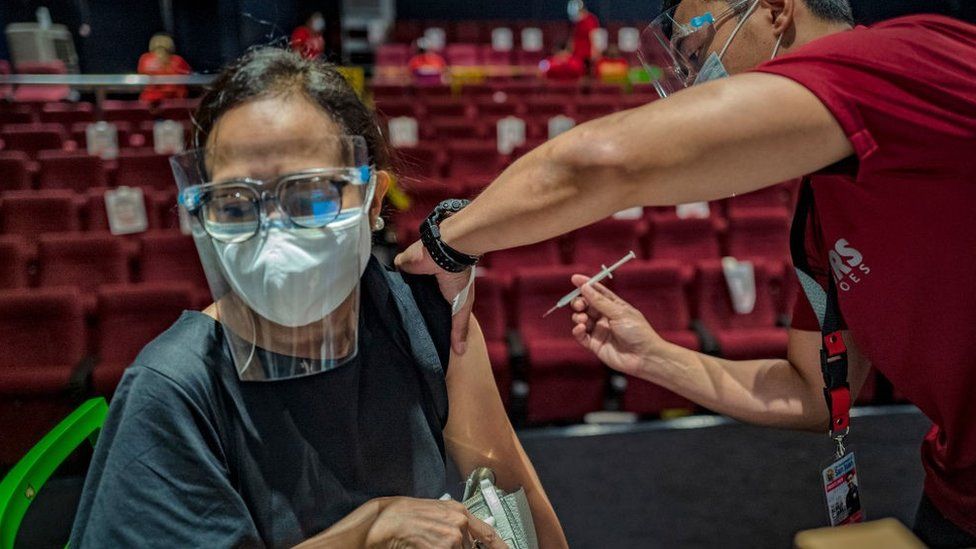
The World Health Organization (WHO) has approved China’s Sinovac Covid vaccine for emergency use.
It is the second Chinese vaccine to receive the green light from the WHO, after Sinopharm.
It opens the door for the jab to be used in the Covax programme, which aims to ensure fair access to vaccines.
The vaccine, which has already been used in several countries, has been recommended for over 18s, with a second dose two to four weeks later.
The emergency approval means the vaccine “meets international standards for safety, efficacy and manufacturing”, the WHO said.
Studies showed that Sinovac prevented symptomatic disease in more than half of those vaccinated and prevented severe symptoms and hospitalization in 100% of those studied, it added.
It is hoped that the decision to list the Chinese vaccine for emergency use will give a boost to the Covax initiative, which has been struggling with supply problems.
“The world desperately needs multiple Covid-19 vaccines to address the huge access inequity across the globe,” said Mariangela Simao, the WHO’s assistant director general for access to health products.
“We urge manufacturers to participate in the Covax facility, share their know-how and data and contribute to bringing the pandemic under control,” she said.
As well as China, the vaccine is already being administered in countries including Chile, Brazil, Indonesia, Mexico, Thailand and Turkey.
Sinovac says it has supplied more than 600 million doses at home and abroad as of the end of May. It says more than 430 million doses have been administered.
One of Sinovac’s main advantages is that it can be stored in a standard refrigerator at 2-8 degrees Celsius. This means Sinovac is a lot more useful to developing countries which might not be able to store large amounts of vaccine at low temperatures.
The emergency approval came as the heads of the WHO, the World Trade Organisation, the International Monetary Fund and the World Bank appealed for a $50bn (£35bn) investment fund to help end the pandemic.
In a joint statement they said the world had reached a perilous point, and that inequalities in access to vaccines risked prolonging the pandemic, and many more deaths.
Source: BBC
You may like
-


Britney Spears’ third marriage hits the rock, split with husband 14 months on
-


Deadly Mediterranean wildfires kill more than 40
-


Ship with 3,000 cars in deadly fire off Dutch coast
-


Ukraine war: Russian general fired after criticising army leaders
-


Tourist submersible visiting wreck 1912 Titanic goes missing in Atlantic Ocean
-


Greece boat disaster: Capsized boat had 100 children in hold



Mahama leads NDC to mourn with Kufuor over wife’s passing
Former President John Dramani Mahama on Wednesday, October 4 led a delegation from the National Democratic Congress (NDC) to visit...


Former First Lady of Ghana Theresa Kufuor reported dead
The former First Lady of Ghana and wife of Ghana’s former President, John Agyekum Kufour, Mrs Theresa Kufuor has been...


The most powerful tool for change is the right to vote – Mahama
The 2024 flagbearer of the National Democratic Congress (NDC), John Dramani Mahama, is rallying Ghanaains who have grown disillusioned with...


PFJ was a mere state resource looting platform – Minority
The Minority has descended heavily on the Akufo-Addo-led government for launching a second phase of the flagship Planting for Food...


ECOWAS is desecrating Ghana
The Director of Legal Affairs of the National Democratic Congress (NDC), Mr Abraham Amaliba has said the Economy Community of...


Prof Gyampo elected President of UTAG, University of Ghana branch
A Senior Lecturer at the Political Science Department of the University of Ghana (Legon), Prof Ransford Gyampo have been elected...
